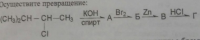timur2312
Пользователи-
Постов
158 -
Зарегистрирован
-
Посещение
Тип контента
Профили
Форумы
События
Весь контент timur2312
-
получите всеми известными способами этилбензол. напишите уравнение реакции его окисления
-
напишите уравнение альдольной конденсации пропаналя
-
При пропускании смеси этана и этина через склянку с бромной водой вес склянки увеличился на 1,3 г, а при сгорании такого же количества смеси углеводородов выделилось 14 л двуокиси углерода (н.у.). Определите объем исходной смеси газов (н.у.).
-
-
подскажите, как назвать данное соединение? Ph3P=NC6H4Cl -3
-
помогите кто-нибудь пожалуйста перевести Selective reduction of the nitro group in 8 presented another major challenge. Conditions such as Zn,1 Zn/HC02NH4, Zn/H2NNH2.HC02H,17 In,18 In/NH4C1,19 Fe,20 Fe-FeCl*21 Mg/H2NNH2-HC02H,22 and SnCl^ as well as Adam's and Lindlar’s catalysts, either led to over-reduction or to no reaction. Transfer hydrogenation using HC02H or H2 over 1096 Pd/C led to over-reduction. On the other hand, use of HC02NH4 with either 10% Pd/C23 or RaNi returned the starting material. Remarkably, HC02H with catalytic RaNi gave diastereomeric amine 9 in 88% yield (Figure 7, structure by X-ray). Conversion of amine 9 into racemic core 10 (D-ring) was accomplished by an intramolecular reductive amination between Cl6 and the nitrogen via in situ hydrolysis of the acetal group using 0.5 N aqueous HC1 at 100 °C followed by formation of an iminium ion and its reduction with NaCNBH3 into a racemic mixture of amines 10 (8,14dihydronorsalutar- idine and norisosinomenine). No hydrolysis of the methyl enol ether occurred under these conditions. A yield of 70% was obtained for 10 which upon treatment with 37% H2CO(aq), NaCNBH3, and AcOH in EtOH25 led to N-methylation to give 11 as a racemic mixture of isosinomenine and 8,14* dihydrosalutaridine in 91% yield (Figure 8, structure by X-ray). The strategy presented here is an efficient (17% from 1, 10 steps) racemic synthesis of the aforementioned 8,14-dihydro- morphinandienone alkaloids. The synthesis furnishes the correct oxidation level of the target compounds by avoiding the biomimetic strategy of o,p-phenolic coupling. A unique route to a Henry—Michael—dehydration cascade has been developed where a variety of other conditions failed. The resultant aliphatic nitro group could be reduced selectively only under transfer hydrogenation conditions. The current strategy is complementary to that from our group for the synthesis of codeine9 in that it begins with the C6 oxygen already in place, obviating the need for its later introduction over several steps. A one-step conversion of 7 to 9, a one-pot conversion of 9 to 11, and an asymmetrical conversion of 5 to 6 are topics for future consideration. Conversion of 8,14-dihydrosalutaridine into salutaridine (i.e., oxidation of C8—C14 into a double bond as exemplified by conversion of ocobotrine into sinoacutine via cr-bromination-dehydrobromination a) would allow for its subsequent transformation into thebaine26 and codeine,27 providing a practical synthesis of these iconic structures.
-
не подскажете как диссоциирует коллоидный раствор KFe[Fe(CN)6]3?
-
специалисты только у нас, за границей учат только бакалавров и магистров, так что специалистов туда не берут степень магистра выше, чем специалиста, а в аспирантуру можно поступить и с дипломом специалиста, но только в россии
-
по диаграмме плавкости для системы SrO- SiO2, установите формулу химического соединения. Рассчитайте, сколько граммов этого соединения содержится в 1,3 кг сплава, содержащего 60% SrO
-
подскажите 3 способа
-
помогите решить Вычислите при 298К ЭДС элемента, используя справочные данные о ПР малорастворимых соединений. Zn|ZnS(насыщенный раствор)||Zn(OH)2(насыщенный раствор)|Zn
-
1) между раствором гемоглобина в буфере и буферным раствором образуется резкая граница при 25 градусах. После 10 ч полуширина кривой концентрация - градиент в точках перегиба равна 0,226 см. Чему равен коэффициент диффузии в этих условиях? 2) Белок плазмы крови человека имеет молекулярный вес 69000. Расчитать осмотическое давление раствора, содержащего 2 г этого белка в 100 см3 при 25 0C в мм рт. ст. и в мм вод. ст.
-
Составить цепочку превращений. из пропионата натрия получить бутин-2.
-
Составьте электронно-ионные схемы и закончите уравнения следующих реакций, определите окислитель и восстановитель и молярную массу эквивалента окислителя и восстановителя FeSO 4+H2O2+H2SO4-> KBrO3+H2SO4+KI->
-
Составьте электронно-ионные схемы и закончите уравнения следующих реакций, определите окислитель и восстановитель и молярную массу эквивалента окислителя и восстановителя FeSO 4+H2O2+H2SO4-> KBrO3+H2SO4+KI->
-
1) Когда можно считать, что тепловой эффект реакции прямо пропорционален Т? 2)что больше для реакции 4NO+6H2O(г)=4NH3+5O2 при 150 0С: Qv или Qp? Объясните
-
Стандартная энтальпия образования формальдегида при 25С равна -108,6 кДж/моль. Рассчитайте стандартную энтальпию образования формальдегида при 150С, если известны теплоемкости: (-110,7 кДж моль-1) Вещество С(графит) Н2(г) N2(г) С2Н5N(г) Ср,Джмоль-1К-1 8,53 28,82 29,13 53,10
-
с какими из перечисленных веществ будет взаимодействовать уксусная кислота? железом, оксидом фосфора (V) сульфидом железа (II) гидроксидом натрия, ртутью, нитратом бария оксидом алюминия приведите уравнения возможных реакций в молекулярном и ионном виде